The Global Minimally Invasive Surgical Systems Market Size, Share & Growth Analysis By Device Type (Handheld Devices, Cutter Devices, Inflation Devices, Electrosurgical Devices, Monitoring and Visual Devices, Guiding Devices, Auxiliary Devices), By Surgery Type (Gastrointestinal Surgery, Cardiovascular Surgery, Orthopedic Surgery, Urological Surgery, Cosmetic Surgery, Neurological Surgery), By End-Use (Hospitals, Specialty Clinics, Ambulatory Surgical Centers), And Regional Forecast, 2021-2027, This research & consulting report doles out information about the global Minimally Invasive Surgical Systems market. The forecast period of the market extends till 2027, while commencing in 2020. The report sheds light on various key insights & trends that have occupied center stage & drawn the attention of end-users. The Minimally Invasive Surgical Systems industry is witnessing impressive expansion at present, having logged double digit CAGRs in the past.
The report alludes to the Minimally Invasive Surgical Systems market as a lucrative sector. This sector is slated to generate substantial revenues during forecast period 2021 to 2027. The analytical report highlights data & overview regarding the size, shares, & demand of Minimally Invasive Surgical Systems. Several other factors have had a positive or negative impact on the industry, all of which have been laid out in details.
The said factors comprise drivers, novel prospects, hindrances, and other vital characteristics of the market. Investments by rivals into Minimally Invasive Surgical Systems products have catapulted the market into limelight & have stoked its demand, globally.
Few among other key aspects that find a mention; encompass the industry segments, predictions, & size and shares for 2021 to 2027. Besides these; the report narrows down on the innovative trends & technological developments, expansion strategies of key players, and regional statistics.
request a free sample copy
The manner in which nascent & local competitors stack up against prominent players, hogs the spotlight. Novel products & services, coupled with the monetary shares of eminent competitors and the SWOT analysis unveil the complexities of the global industry size.
This report forecasts revenue growth at the global, regional, and country levels and provides an analysis of the latest industry trends from 2021 to 2027 in each of the sub segments. For the purpose of this study, Eternity Insights has segmented the global Minimally Invasive Surgical Systems market on the basis of device type, surgery type, end-use and region:
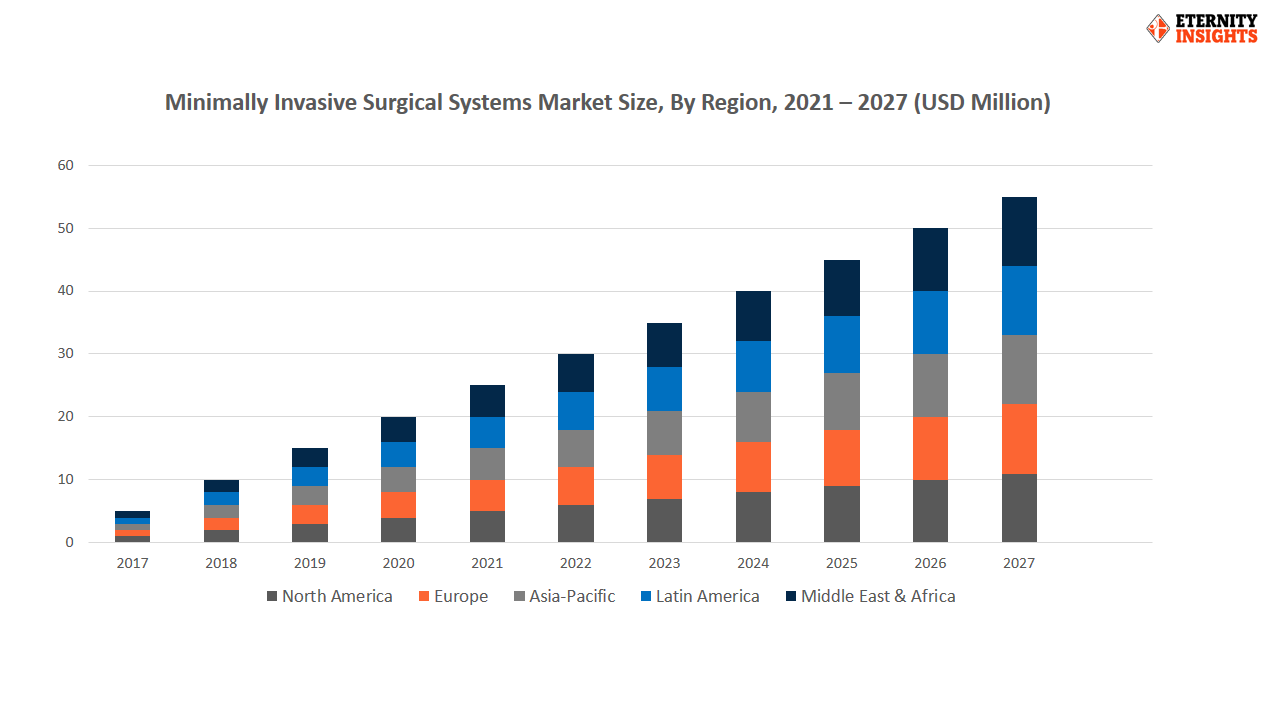
To learn more about this report, request a free sample copy
Research methodology is an integral component of any kind of high-level market research. It entails evaluation by expert panels, research on secondary & primary levels, checks on quality, & testing on completion. Secondary research deeply focuses on the whys, whos, wheres, whens, & hows and caters to answering the strictly consumer-centric base of people.
Some other secondary research sources comprise governmental affiliations & blogs, business magazines, and industrial journals. Primary research includes face-to-face & telephonic interviews with industry experts. It is comparatively more detailed & lays stress on unprejudiced opinions about the products & services.
Primary research findings cement secondary research & help end-users & businesses get a better hang of various markets. It is a well-established fact, though, that research methodology is best implemented through the mixed execution of its aforementioned procedures.
What Does the Report Offer?
Other Vital Points That Are Covered
The market report also brings to light major opportunities and untapped geographies. It is more like a blend of customized research documents & conventional reports. Besides informing buyers about the pros & cons of direct and indirect sales channels, the analysis exhibits illustrations & graphical representations of industry growth and the innovative trends within the industry.
1 Product Definition
2 Scope of Research
2.1 Research Scope
2.2 Key Questions Answered in the Report
3 Research Methodology
3.1 Primary Research
3.2 Secondary Research
3.3 Data Sources Categorization
3.4 Companies Profiled in the Report
3.5 Brief Overview of the Market Estimation Process
3.6 Data Validation
3.7 Assumptions and Limitations
4 Industry Analysis
4.1 Industry Structure
4.1.1 Conventional Minimally Invasive Surgical Systems Manufacturers
4.1.2 Surgical Robotic Systems Vendors
4.1.3 Contract Manufacturers
4.1.4 Distributors
4.2 Industry Supply Chain Analysis
4.3 Regulatory Framework and Government Initiatives
4.3.1 Regulatory Pathway for Minimally Invasive Surgical Systems in the U.S.
4.3.2 Regulations in the European Union (EU)
4.3.2.1 Impact of the EU’s Medical Device Regulations (MDR)
4.3.2.1.1 Expansion of Product Scope
4.3.2.1.2 Reclassification of Medical Devices as per Associated Risk, Contact Duration, and Invasiveness
4.3.2.1.3 More Supportive Clinical Investigations for Class III and Implantable Medical Devices
4.3.2.1.4 Dedicated Personnel for MDR Compliance
4.3.2.1.5 More Emphasis on Post-Market Surveillance
4.3.2.1.6 Requirement of Common Specifications
4.3.2.1.7 Implementation of a Unique Device Identification (UDI) Mechanism
4.3.3 Regulations in Other Countries
4.3.4 Consortiums and Regulatory Bodies
4.4 Patent Analysis
4.4.1 Awaited Technology Developments
4.4.1.1 The Advent of Miniature Minimally Invasive Surgical Instruments
4.4.1.2 Advancement in Haptic Feedback
4.4.1.3 The Age of AI-Integration
4.4.2 Patent Filing Trend
4.4.2.1 Surgical Robotics
4.4.2.2 Conventional MISS
4.4.3 Patent Expiration Impact
4.5 Industry Opportunity Analysis
5 Competitive Landscape
5.1 Market Share Analysis
5.1.1 Surgical Robotics
5.1.2 Conventional MISS
5.2 Key Strategies and Developments
5.2.1 Conventional Minimally Invasive Surgical Systems
5.2.1.1 Funding Activities
5.2.1.2 M&A Activities
5.2.1.3 Partnerships, Alliances and Business Expansion
5.2.1.4 Regulatory and Legal
5.2.1.5 New Offerings
5.2.2 Surgical Robotics
5.2.2.1 Funding Activities
5.2.2.2 M&A Activities
5.2.2.3 Partnerships, Alliances, and Business Expansions
5.2.2.4 Regulatory and Legal
5.2.2.5 New Offerings
5.3 Business Model Analysis
6 Global Minimally Invasive Surgical Systems Market Scenario
6.1 Assumptions and Limitations
6.2 Key Findings and Opportunity Assessment
6.3 Global Minimally Invasive Surgical Systems Market Size and Forecast
6.4 Market Dynamics
6.4.1 Conventional Minimally Invasive Surgical Systems
6.4.1.1 Impact Analysis
6.4.1.2 Market Drivers
6.4.1.2.1 Rise in the Number of Chronic Diseases
6.4.1.2.2 Advantages Over Open Surgeries
6.4.1.2.3 Increase in Global Geriatric Population
6.4.1.2.4 Increasing Healthcare Costs
6.4.1.2.5 Technological Advances in Surgeries
6.4.1.2.6 Growth in Number of Surgical Procedures
6.4.1.3 Market Restraints
6.4.1.3.1 Availability of Resources Required for MIS Procedures
6.4.1.3.2 Shortage of Skilled Professionals
6.4.1.3.3 Restrictive Reimbursement Landscape
6.4.1.4 Market Opportunities
6.4.1.4.1 Engage in Mergers and Acquisitions to Diversify Endoscopic Portfolio
6.4.1.4.2 Invest in Regional Expansion
6.4.2 Surgical Robotics
6.4.2.1 Impact Analysis
6.4.2.2 Market Drivers
6.4.2.2.1 Rising Incidence of Chronic Disorders
6.4.2.2.2 Growth in Geriatric Population
6.4.2.2.3 Increase in Healthcare Spending
6.4.2.2.4 Technological Advancement in the Field of Medical Surgeries
6.4.2.3 Market Restraints
6.4.2.3.1 High Cost of Surgical Robotic Systems
6.4.2.3.2 Shortage of Skilled Professionals
6.4.2.3.3 Restrictive Reimbursement Landscape
6.4.2.4 Market Opportunities
6.4.2.4.1 Development of Low-Cost Surgical Robotic Systems
6.4.2.4.2 Development of Micro- and Nano-Robots for Surgeries
6.4.2.4.3 Implementation of Artificial Intelligence (AI) in Robotics
6.4.2.4.4 Development of Surgical Simulators for the Training of Professionals
6.5 Market Estimation Method
6.6 Key Vendors
7 Global Minimally Invasive Surgical Systems Market (by Product Type), 2017-2027
7.1 Key Findings and Opportunity Assessment
7.2 Conventional Minimally Invasive Surgical Systems (MISS)
7.2.1 Endoscopes and Endoscopic Systems
7.2.1.1 Rigid Endoscopes
7.2.1.1.1 Urology Rigid Endoscopes
7.2.1.1.1.1 Cystoscopes
7.2.1.1.1.2 Resectoscopes
7.2.1.1.1.3 Nephroscopes
7.2.1.1.1.4 Other Urology Endoscopes
7.2.1.1.2 Laparoscopes
7.2.1.1.3 Arthroscopes
7.2.1.1.4 Gynecology Endoscopes
7.2.1.1.5 Neuroendoscopes
7.2.1.1.6 ENT Endoscopes
7.2.1.1.7 Other Rigid Endoscopes
7.2.1.2 Flexible Endoscopes
7.2.1.2.1 Gastrointestinal Flexible Endoscopes
7.2.1.2.1.1 Gastroscopes
7.2.1.2.1.2 Duodenoscopes
7.2.1.2.1.3 Enteroscopes
7.2.1.2.1.4 Colonoscopes
7.2.1.2.1.5 Sigmoidoscopes
7.2.1.2.1.6 Other GI Endoscopes
7.2.1.2.2 Urology Flexible Endoscopes
7.2.1.2.3 Gynecology Flexible Endoscopes
7.2.1.2.4 Bronchoscopes
7.2.1.2.5 Laryngoscopes
7.2.1.2.6 Pharyngoscopes
7.2.1.2.7 ENT Endoscopes
7.2.1.3 Capsule Endoscopy
7.2.1.3.1 Workstations and Data Recorders
7.2.1.3.2 Capsules
7.2.1.3.3 Others (Services and Patency System)
7.2.2 Video and Visualization Systems
7.2.2.1 Cameras
7.2.2.1.1 Single-Chip Cameras
7.2.2.1.2 3-Chip Cameras
7.2.2.2 Displays and Monitors
7.2.2.2.1 Wireless Displays and Monitors
7.2.2.2.2 Wired Displays and Monitors
7.2.2.3 Video Recorders
7.2.2.3.1 Standalone
7.2.2.3.2 Inbuilt
7.2.2.4 Camera Heads
7.2.2.4.1 Charged Couple Device (CCD)
7.2.2.4.2 Complementary Metal Oxide Semiconductor (CMOS)
7.2.2.5 Light Sources
7.2.2.5.1 LED
7.2.2.5.2 Xenon
7.2.2.5.3 Laser
7.2.2.5.4 Halogen
7.2.2.6 Video Converters
7.2.2.7 Video Processors
7.2.2.8 Printers
7.2.3 Instruments and Accessories
7.2.3.1 Insufflators
7.2.3.2 Irrigation Systems
7.2.3.3 Endoscope Tip Protector, Tubing and Water Systems
7.2.3.4 Procedure Kits
7.2.3.5 Forceps
7.2.3.6 Retrieval Kits
7.2.3.7 Injection Needles
7.2.3.8 Energy Devices
7.2.3.9 Closure Devices
7.2.3.10 Hand Instruments
7.2.3.11 Access Devices
7.2.3.12 Others
7.2.4 Others
7.3 Surgical Robotics
7.3.1 Robotic Systems
7.3.2 Robotic Surgery Instruments and Accessories
7.3.3 Services
8 Global Minimally Invasive Surgical Systems Market (by Application), 2017-2027
8.1 Key Findings and Opportunity Assessment
8.2 Conventional Minimally Invasive Surgical Systems Market Share (by Application), 2017-2027
8.2.1 Conventional Minimally Invasive General Surgery
8.2.2 Conventional Minimally Invasive Urology Surgery
8.2.3 Conventional Minimally Invasive Gynecology Surgery
8.2.4 Conventional Minimally Invasive Orthopedic Surgery
8.2.5 Conventional Minimally Invasive Cardiovascular Surgery
8.2.6 Conventional Minimally Invasive Neurosurgery
8.2.7 Others
8.3 Surgical Robotics Market Share (by Application), 2017-2027
8.3.1 Robotic-Assisted General Surgery
8.3.2 Robotic-Assisted Urology Surgery
8.3.3 Robotic-Assisted Gynecology Surgery
8.3.4 Robotic-Assisted Orthopedic Surgery
8.3.5 Robotic-Assisted Cardiovascular Surgery
8.3.6 Robotic-Assisted Neurosurgery
8.3.7 Others
9 Global Minimally Invasive Surgical Systems Market (by End User), 2017-2027
9.1 Key Findings and Opportunity Assessment
9.2 Conventional Minimally Invasive Surgical Systems Market Share (by End User), 2017-2027
9.2.1 Hospitals
9.2.2 Ambulatory Surgical Centers
9.2.3 Specialty Clinics
9.3 Surgical Robotics Market Share (by End User), 2017-2027
9.3.1 Hospitals
9.3.2 Ambulatory Surgical Centers
9.3.3 Specialty Clinics
10 Global Minimally Invasive Surgical Systems Market (by Region)
10.1 Overview
10.2 North America Minimally Invasive Surgical Systems Market
10.2.1 Key Findings and Opportunity Assessment
10.2.2 Market Size and Forecast
10.2.3 Market Dynamics
10.2.4 Key Vendors
10.2.5 U.S.
10.2.5.1 U.S. Minimally Invasive Surgical Analysis (by Product Type)
10.2.6 Canada
10.2.6.1 Canada Minimally Invasive Surgical Analysis (by Product Type)
10.3 Europe Minimally Invasive Surgical Systems Market
10.3.1 Key Findings and Opportunity Assessment
10.3.2 Market Size and Forecast
10.3.3 Market Dynamics
10.3.4 Key Vendors
10.3.5 Germany
10.3.5.1 Germany Minimally Invasive Surgical Analysis (by Product Type)
10.3.6 France
10.3.6.1 France Minimally Invasive Surgical Analysis (by Product Type)
10.3.7 Italy
10.3.7.1 Italy Minimally Invasive Surgical Analysis (by Product Type)
10.3.8 U.K.
10.3.8.1 U.K. Minimally Invasive Surgical Analysis (by Product Type)
10.3.9 Spain
10.3.9.1 Spain Minimally Invasive Surgical Analysis (by Product Type)
10.3.10 Belgium
10.3.10.1 Belgium Minimally Invasive Surgical Analysis (by Product Type)
10.3.11 Switzerland
10.3.11.1 Switzerland Minimally Invasive Surgical Analysis (by Product Type)
10.3.12 Sweden
10.3.12.1 Sweden Minimally Invasive Surgical Analysis (by Product Type)
10.3.13 Netherlands
10.3.13.1 Netherlands Minimally Invasive Surgical Analysis (by Product Type)
10.3.14 Denmark
10.3.14.1 Denmark Minimally Invasive Surgical Analysis (by Product Type)
10.3.15 Rest-of-Europe
10.3.15.1 Rest-of-Europe Minimally Invasive Surgical Analysis (by Product Type)
10.4 Asia-Pacific Minimally Invasive Surgical Systems Market
10.4.1 Key Findings and Opportunity Assessment
10.4.2 Market Size and Forecast
10.4.3 Market Dynamics
10.4.4 Key Vendors
10.4.5 Japan
10.4.5.1 Japan Minimally Invasive Surgical Analysis (by Product Type)
10.4.6 China
10.4.6.1 China Minimally Invasive Surgical Analysis (by Product Type)
10.4.7 India
10.4.7.1 India Minimally Invasive Surgical Analysis (by Product Type)
10.4.8 South Korea
10.4.8.1 South Korea Minimally Invasive Surgical Analysis (by Product Type)
10.4.9 Australia and New Zealand
10.4.9.1 Australia and New Zealand Minimally Invasive Surgical Analysis (by Product Type)
10.4.10 Malaysia
10.4.10.1 Malaysia Minimally Invasive Surgical Analysis (by Product Type)
10.4.11 Singapore
10.4.11.1 Singapore Minimally Invasive Surgical Analysis (by Product Type)
10.4.12 Rest-of-Asia-Pacific
10.4.12.1 Rest-of-Asia-Pacific Minimally Invasive Surgical Analysis (by Product Type)
10.5 Latin America Minimally Invasive Surgical Systems Market
10.5.1 Key Findings and Opportunity Assessment
10.5.2 Market Size and Forecast
10.5.3 Market Dynamics
10.5.4 Key Vendors
10.5.5 Brazil
10.5.5.1 Brazil Minimally Invasive Surgical Analysis (by Product Type)
10.5.6 Mexico
10.5.6.1 Mexico Minimally Invasive Surgical Analysis (by Product Type)
10.5.7 Argentina
10.5.7.1 Argentina Minimally Invasive Surgical Analysis (by Product Type)
10.5.8 Rest-of-Latin America
10.5.8.1 Rest-of-Latin America Minimally Invasive Surgical Analysis (by Product Type)
10.6 Middle East and Africa Minimally Invasive Surgical Systems Market
10.6.1 Key Findings and Opportunity Assessment
10.6.2 Market Size and Forecast
10.6.3 Market Dynamics
10.6.4 Key Vendors
10.6.5 Kingdom of Saudi Arabia (K.S.A.)
10.6.5.1 K.S.A. Minimally Invasive Surgical Analysis (by Product Type)
10.6.6 Israel
10.6.6.1 Israel Minimally Invasive Surgical Analysis (by Product Type)
10.6.7 U.A.E.
10.6.7.1 U.A.E. Minimally Invasive Surgical Analysis (by Product Type)
10.6.8 Rest-of-MEA
10.6.8.1 Rest-of-MEA Minimally Invasive Surgical Analysis (by Product Type)
11 Company Profiles
11.1 Overview
11.2 Auris Health, Inc. (Johnson & Johnson)
11.2.1 Company Overview
11.2.2 Auris Health, Inc.: Company Description
11.2.3 Auris Health, Inc.: SWOT Analysis
11.3 Boston Scientific Corporation
11.3.1 Company Overview
11.3.2 Boston Scientific Corporation: Company Description
11.3.3 Financials
11.3.4 Boston Scientific Corporation: SWOT Analysis
11.4 Cook Medical
11.4.1 Company Overview
11.4.2 Cook Medical: Company Description
11.4.3 Cook Medical: SWOT Analysis
11.5 Corindus Vascular Robotics, Inc.
11.5.1 Company Overview
11.5.2 Corindus Vascular Robotics, Inc.: Company Description
11.5.3 Financials
11.5.4 Corindus Vascular Robotics, Inc.: SWOT Analysis
11.6 Intuitive Surgical, Inc.
11.6.1 Company Overview
11.6.2 Intuitive Surgical, Inc.: Company Description
11.6.3 Financials
11.6.4 Intuitive Surgical Inc.: SWOT Analysis
11.7 Medrobotics Corporation
11.7.1 Company Overview
11.7.2 Medrobotics Corporation: Company Description
11.7.3 Medrobotics Corporation: SWOT Analysis
11.8 Medtech SA (Zimmer Biomet Holdings, Inc.)
11.8.1 Company Overview
11.8.2 Zimmer Biomet Holdings, Inc.: Company Description
11.8.3 Financials
11.8.4 Zimmer Biomet Holdings, Inc.: SWOT Analysis
11.9 Mazor Robotics Ltd. (Medtronic plc)
11.9.1 Company Overview
11.9.2 Mazor Robotics Ltd.: Company Description
11.9.3 Financials
11.9.4 Mazor Robotics Ltd.: SWOT Analysis
11.10 Olympus Corporation
11.10.1 Company Overview
11.10.2 Olympus Corporation: Company Description
11.10.3 Financials
11.10.4 Olympus Corporation: SWOT Analysis
11.11 Renishaw plc
11.11.1 Company Overview
11.11.2 Renishaw Plc: Company Description
11.11.3 Financials
11.11.4 Renishaw plc: SWOT Analysis
11.12 Restoration Robotics, Inc.
11.12.1 Company Overview
11.12.2 Restoration Robotics, Inc.: Company Description
11.12.3 Financials
11.12.4 Restoration Robotics, Inc.: SWOT Analysis
11.13 Smith & Nephew plc
11.13.1 Company Overview
11.13.2 Smith & Nephew plc: Company Description
11.13.3 Financials
11.13.4 Smith & Nephew plc: SWOT Analysis
11.14 Stereotaxis, Inc.
11.14.1 Company Overview
11.14.2 Stereotaxis, Inc.: Company Description
11.14.3 Financials
11.14.4 Stereotaxis, Inc.: SWOT Analysis
11.15 Mako Surgical Corp. (Stryker Corporation)
11.15.1 Company Overview
11.15.2 Mako Surgical Corp. (Stryker Corporation): Company Description
11.15.3 Financials
11.15.4 Stryker Corporation: SWOT Analysis
11.16 THINK Surgical, Inc.
11.16.1 Company Overview
11.16.2 THINK Surgical, Inc.: Company Description
11.16.3 THINK Surgical, Inc.: SWOT Analysis
11.17 TransEnterix, Inc.
11.17.1 Company Overview
11.17.2 TransEnterix, Inc.: Company Description
11.17.3 Financials
11.17.4 TransEnterix, Inc.: SWOT Analysis
To ensure high-level data integrity, accurate analysis, and impeccable forecasts
For complete satisfaction
On-demand customization of scope of the report to exactly meet your needs
Targeted market view to provide pertinent information and save time of readers
A faster and efficient way to cater to the needs with continuous iteration


Focus on Data Accuracy & Reliability

75+ Clients in Fortune 500

All your transactions are secured end-to-end, ensuring a satisfactory purchase

Ensure the best and affordable pricing







